Overall compliance
92.0%
+5.2% vs goal
Diary completion (90 days)
95.0%
2–3× vs baseline
Compliance & retention for modern trials
Delve Health blends eCOA, human concierge, and digital measures so your trials hit compliance and retention targets without adding burden for sites.
92–98% diary completion · 2–3× retention uplift.
Overall compliance
92.0%
+5.2% vs goal
Diary completion (90 days)
95.0%
2–3× vs baseline
Compliance & retention for modern trials
Delve Health blends eCOA, human concierge, and digital measures so your trials hit compliance and retention targets without adding burden for sites.
92–98% diary completion · 2–3× retention uplift.
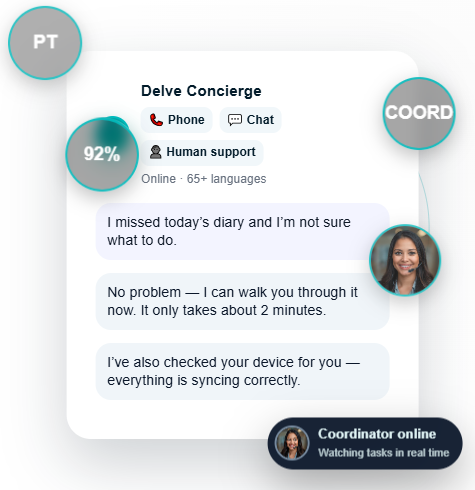
Delve Concierge
Online · 65+ languages
Outcomes that matter
Reduce dropouts with proactive, human-led follow-up and real-time alerts.
Increase compliance with multilingual support and personalized patient journeys.
Run hybrid and decentralized trials without adding vendors or complexity.
Access real-time data and analytics to make faster, better-informed decisions.
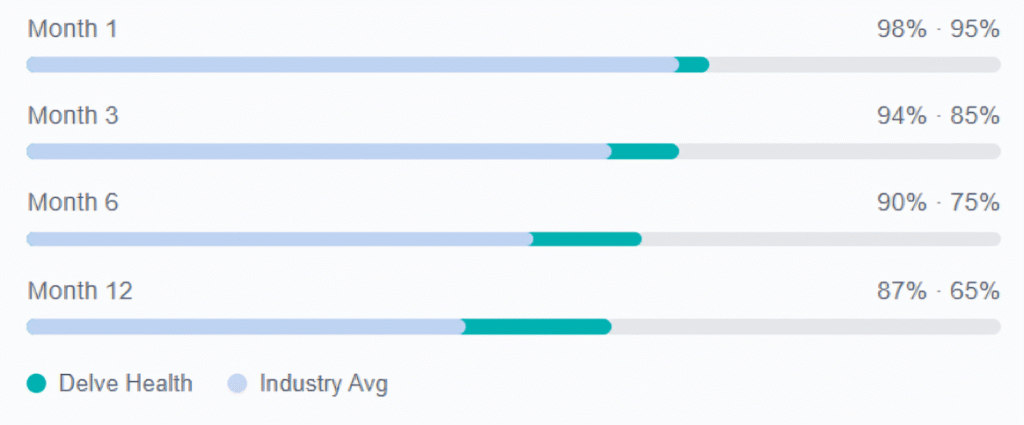
Delve Health vs. industry average
Outcomes that matter
Reduce dropouts with proactive, human-led follow-up and real-time alerts.
Increase compliance with multilingual support and personalized patient journeys.
Run hybrid and decentralized trials without adding vendors or complexity.
Access real-time data and analytics to make faster, better-informed decisions.
Delve Health vs. industry average
Trusted by leading pharma, biotech, and medtech teams
eDiary / ePRO Completion
Improvement in Retention
Fewer Missed Tasks & Syncs
Less Site Burden
Human-driven clinical trial
Technology can remind. Humans get results. Delve’s concierges watch study activity in real time and step in the moment a participant drifts—protecting compliance before it becomes a deviation or a dropout.
They also pre-screen referrals, guide patients through structured questions, confirm interest, and deliver only qualified candidates to sites.
The result: higher completion, fewer dropouts, and calmer, more predictable studies.

Real-time monitoring with fast, human follow-up.
Calling referrals and qualifying only eligible, interested patients.
Empathetic outreach in 65+ languages.
We handle reminders, rescheduling, and participant questions.
Delve wraps compliance and retention around an integrated digital platform—combining
eCOA, digital measures, consent, and analytics with a human layer that keeps
everything moving.
Accessible, multilingual diaries and outcomes that patients can actually complete.
Validated, fit-for-purpose measures from wearables and sensors, ready for regulators.
Human teams who own the work of keeping participants active, informed, and on schedule.
Real-time dashboards that surface risk early and prove the health of your trial.
Whether you’re developing a drug, launching a device, or leading a site network, Delve
Health surrounds your study with the compliance and retention support it needs
to finish strong.
Protect pivotal endpoints with high diary completion and retention.
Support post-market and real-world studies with engaged participants.
Give lean teams an experienced compliance partner from day one.
Offload reminders, calls, and rescheduling so staff can focus on care.
Sites reclaim hours every week — and patients stay supported between visits.

Visits reduced 12 → 2. Diary completion > 95%. Retention: 94%.
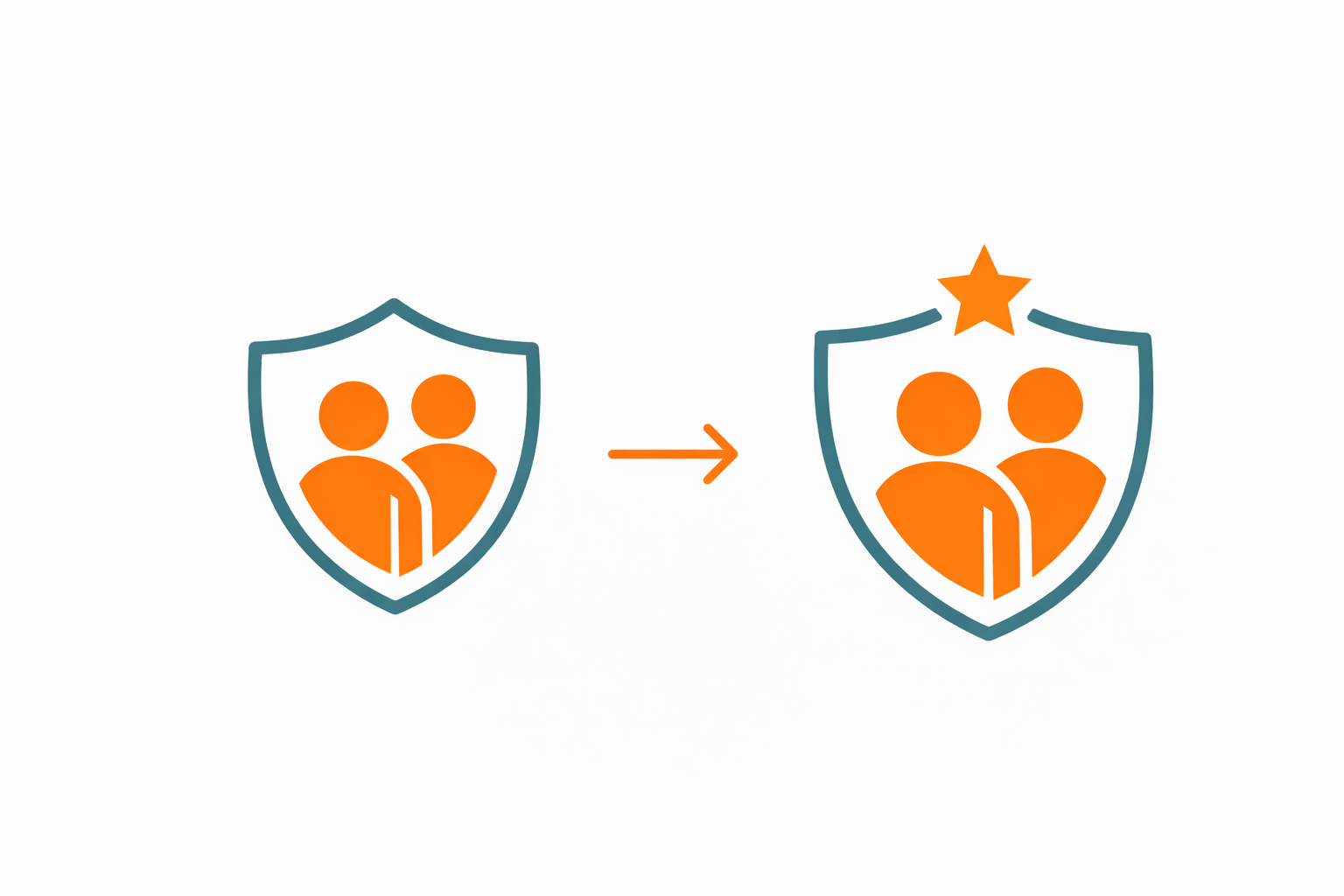
63% increase in retention after layering in concierge support.
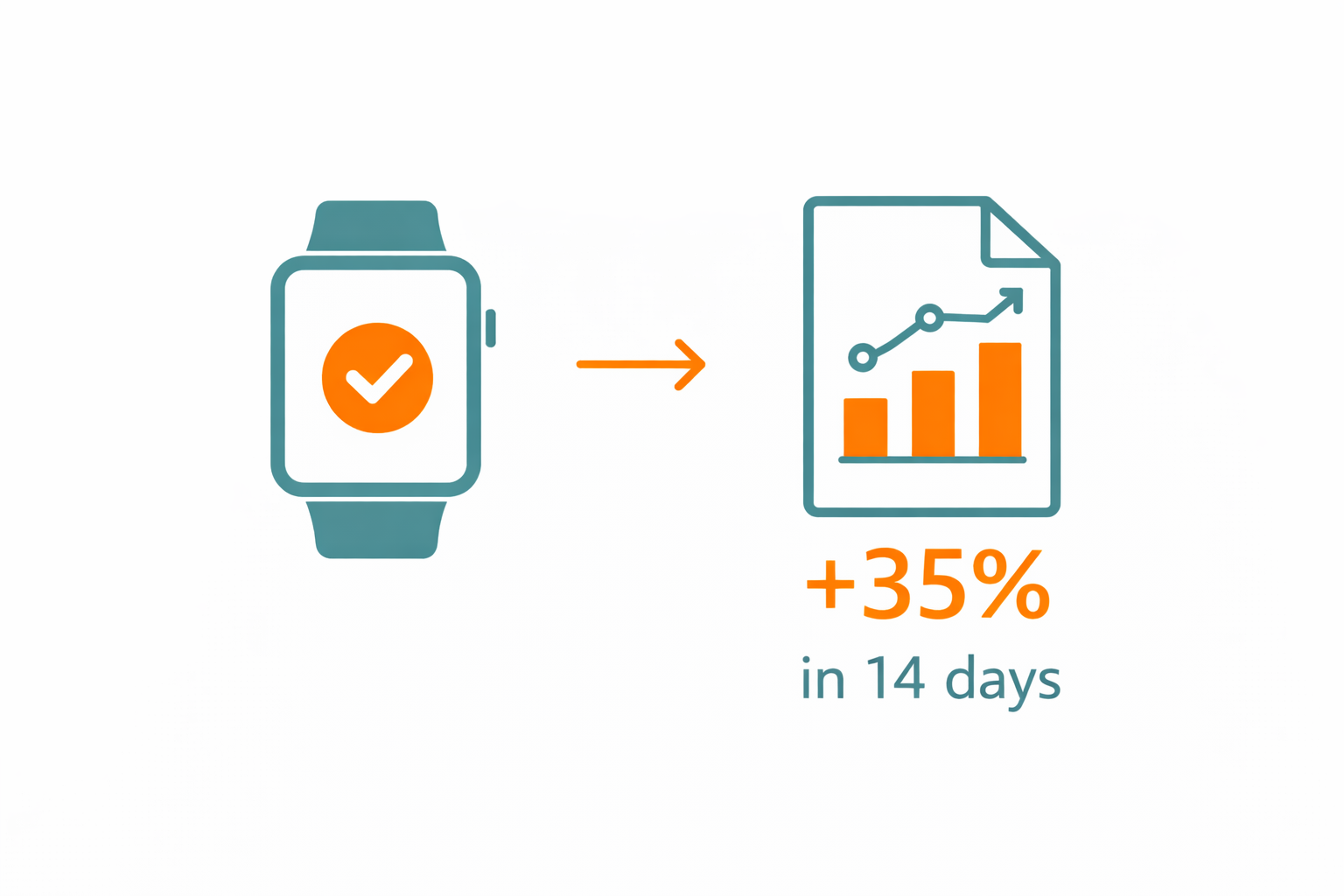
35% increase in usable data within the first 14 days.
Testimonials
Director of Clinical Operations, Global Biotech Sponsor
With Delve, we finally had one place to see compliance, retention, and risk. We completed enrollment three months early with cleaner data at database lock.
James Chen
VP, Clinical Development, Mid-size Pharma
One vendor instead of five simplified our operations dramatically. The concierge services alone were worth the investment.
Maria Rodriguez
Clinical Trial Manager, Leading CRO
Bring Delve into your next protocol as the compliance and retention layer so your team can focus on the science, not the chasing.