Lean operations
Fewer vendors
One platform + one team
Study execution
Faster
Configured and deployed in days
Data quality
Cleaner
Real-time QC + human follow-through
Built for emerging biotech teams
Delve Health helps biotechs run efficient, high-quality decentralized and hybrid studies without massive internal teams or complex multi-vendor setups. Unified data capture, real-time oversight, and human support keep execution on track from first patient to readout.
Less overhead · Faster timelines · More defensible evidence.
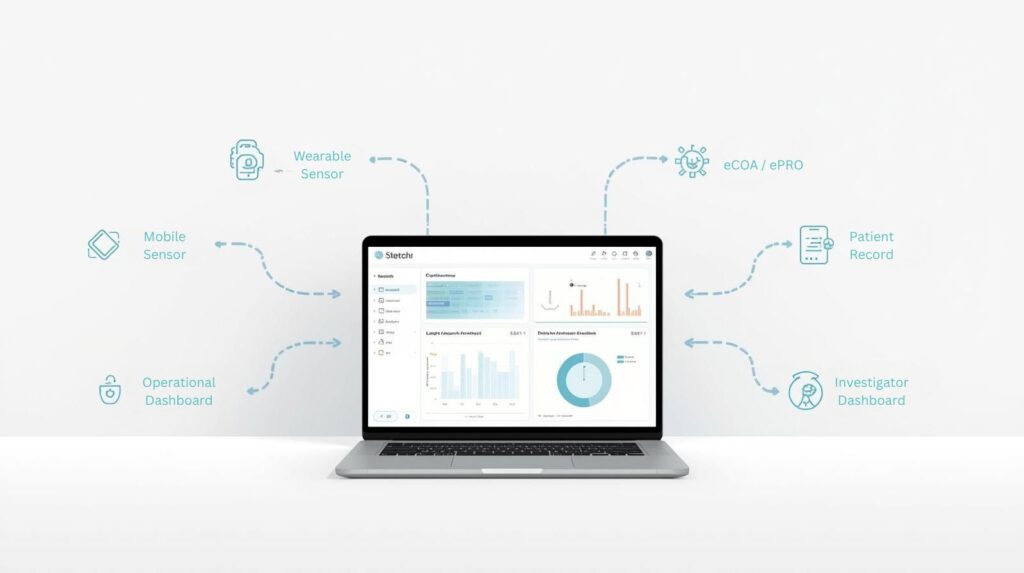
Biotechs need to move quickly, operate lean, and build evidence that stands up to regulatory scrutiny. Delve Health brings together eCOA, wearables, diaries, patient engagement, and concierge support into one platform purpose-built for smaller teams.
Move fast without sacrificing data quality. Delve gives lean teams a unified platform for eCOA,
wearables, and compliance—backed by real humans when execution gets
messy.
Launch eCOA and wearables in weeks, not months.
Validated digital biomarkers aligned with regulatory expectations.
Multilingual outreach keeps participants engaged and compliant.
Automated oversight plus human support reduces operational load.
Early-phase and exploratory studies rely on accurate, continuous data. Delve provides a single, validated infrastructure for collecting both subjective and objective endpoints.

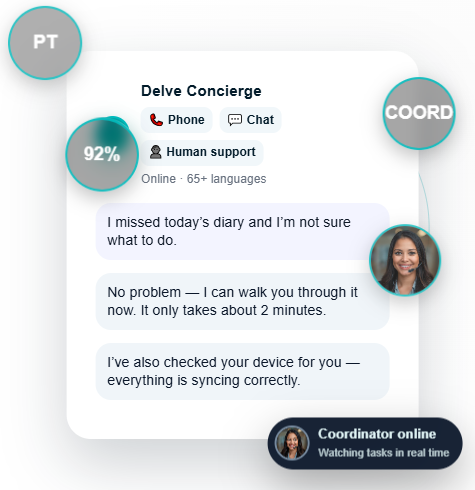
Early-phase studies struggle with variability. When a patient gets stuck, a device misbehaves, or a diary is missed — our concierge intervenes in real time to protect data quality.

Objective signals validated against gold-standard methods.
92% ePRO completion across a hard-to-reach population.
35% faster data readouts with real-time dashboarding.
ePRO Completion
Patient Retention
Wearable Compliance
Faster Interim Readouts
One platform. Validated endpoints. Human retention. Everything a biotech needs to scale evidence.