Comprehension
With multimedia guidance
Consent completion
Faster
Less back-and-forth
Oversight
Real-time
Versioning + audit trail
Consent that patients actually understand
Empower patients with clear, multimedia-guided consent they can understand. Built for global enrollment, hybrid trials, and real-time oversight—without adding burden to sites.
Video + visuals · Comprehension checks · eSignature · Full audit trail.
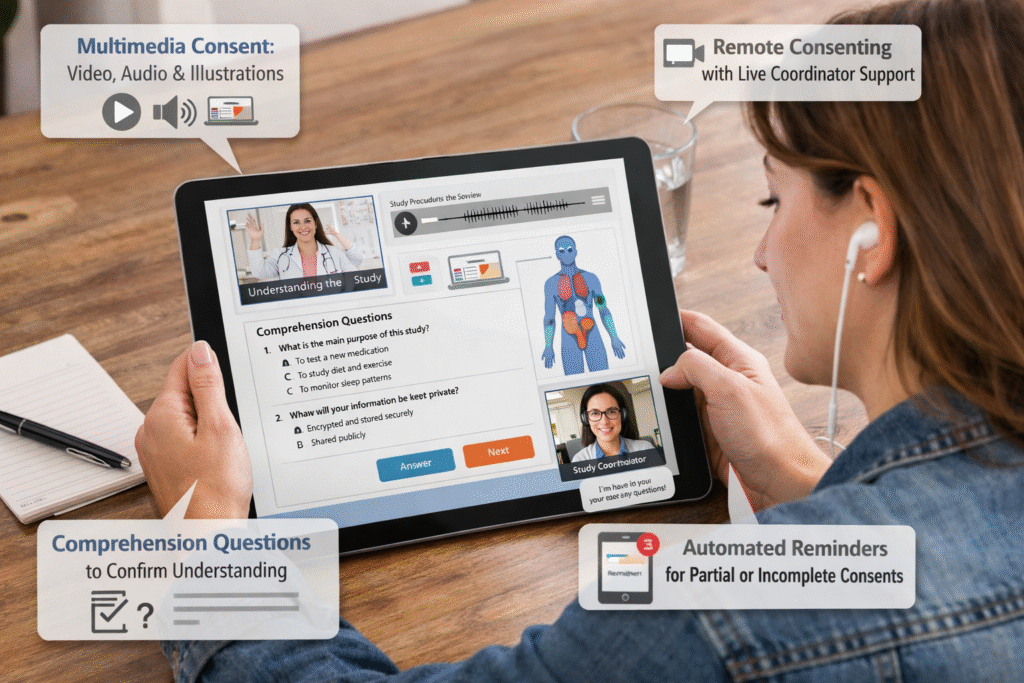
Most eConsent platforms digitize PDFs. Delve rethinks consent entirely — using multimedia, comprehension checks, human support, and real-time site oversight to ensure every participant truly understands the study.
Effective eConsent is more than a signature. It ensures comprehension, regulatory
integrity, and real human support throughout the process.
Replace dense legal text with video, visuals, and voice guidance so participants truly understand what they’re consenting to.
Full audit trails, version control, signature verification, and alignment with FDA, EMA, and ICH E6(R3) guidance.
Concierge teams assist patients during consent—answering questions, resolving issues, and ensuring complete documentation.
Video, audio, images, simplified text, chapter-style navigation. Designed for every literacy level and culture — not just for English-speaking populations.

Consent only matters if patients understand it. Delve adds built-in comprehension checks and robust documentation.

When a patient pauses, drops off, or fails a comprehension quiz, a Delve coordinator reaches out. This reduces delays and increases completion rates.

Live visibility into who started consent, who completed it, who needs follow-up.
Automatic notifications when patients pause or fail to complete a section.
Sites, monitors, and sponsors see only what they need — nothing more.
HIPAA, GDPR, 21 CFR Part 11-aligned consent storage with audit history.
Consent completion rates
Faster screening
Languages supported
Regulatory audit-ready
Multimedia. Human Support. Real-Time Oversight. Everything you need to start patients on the right path.