Patient retention
With human-led follow-up
Data completeness
High
Wearables + ePRO + devices
Regulatory readiness
PMS / PMCF
EU MDR · FDA · Global
Post-market clinical trials & real-world evidence
From post-market surveillance to long-term device performance, Delve Health supports medical device studies with unified data capture, human engagement, and real-time oversight—so evidence is reliable, defensible, and submission-ready.
Designed for post-approval studies, registries, and long-term follow-up.
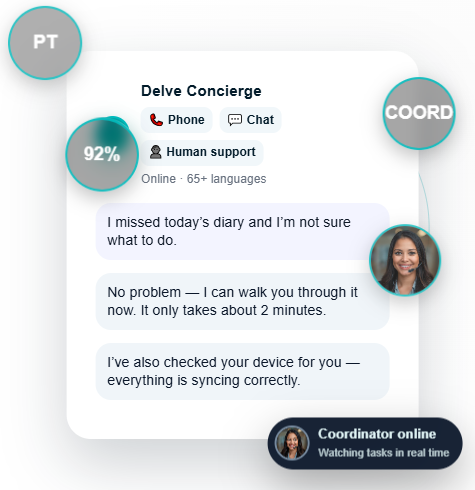
Whether you’re launching a new device, conducting usability studies, or generating real-world performance data, Delve Health gives MedTech teams a unified digital backbone — combining eCOA, wearables, device telemetry, and concierge support into one coordinated workflow.
From post-market follow-up to real-world performance monitoring, Delve supports
medical device teams with validated data, global deployment, and human-backed
compliance.
Rapid deployment for PMCF, vigilance, and real-world evaluations.
Patient and clinician education with reminders, videos, and live support.
Seamless connection to cellular and Bluetooth-enabled sensors.
Monitor device performance, adherence, and signal quality live.
Devices don’t always sync, batteries die, and users make mistakes. That’s why Delve pairs automation with a multilingual human concierge to ensure every device performs — and every dataset stays clean.

Replace complex, multi-vendor setups with a single ecosystem that brings together symptom diaries, device telemetry, video training, visit reminders, and regulatory audit trails.


Visits reduced from 12 → 2 using remote monitoring.

71% improvement in compliance using concierge support.
38% reduction in missing data with automated alerts.
Device Compliance
Retention at 12 Months
Reduction in Sync Failures
Faster Real-World Data Collection
Combine automation, wearables, and real humans in one unified workflow.