Provisioned + shipped
Global
Direct-to-patient or to-site
Replacement workflow
Fast
Lost/damaged device handling
Connectivity
SIM
Cellular-enabled kits available
Operational device management for clinical trials
From sourcing to kitting to enrollment to replacement, Delve manages every device, every SIM, and every update—so sites and sponsors never have to. Standardized kits, documented workflows, and real-time visibility keep study operations moving.
Reduce site burden · Prevent delays · Keep participants connected.
Clinical trials fail when devices fail. Delve handles the entire operational layer — sourcing, configuring, shipping, tracking, updating, and recovering devices across global deployments.

Device programs fail when provisioning is inconsistent, connectivity breaks, or returns
become chaos. Delve runs logistics like a regulated operation—repeatable, auditable,
and global.
Global shipping, customs handling, local carriers, and managed returns.
Apps installed, restrictions applied, wearables paired and validated.
SIM activation, carrier selection, APN setup, and signal validation.
NIST 800-88 certified data deletion, refurbishment, and re-kitting.
Countries Supported
Device Uptime
Devices Provisioned
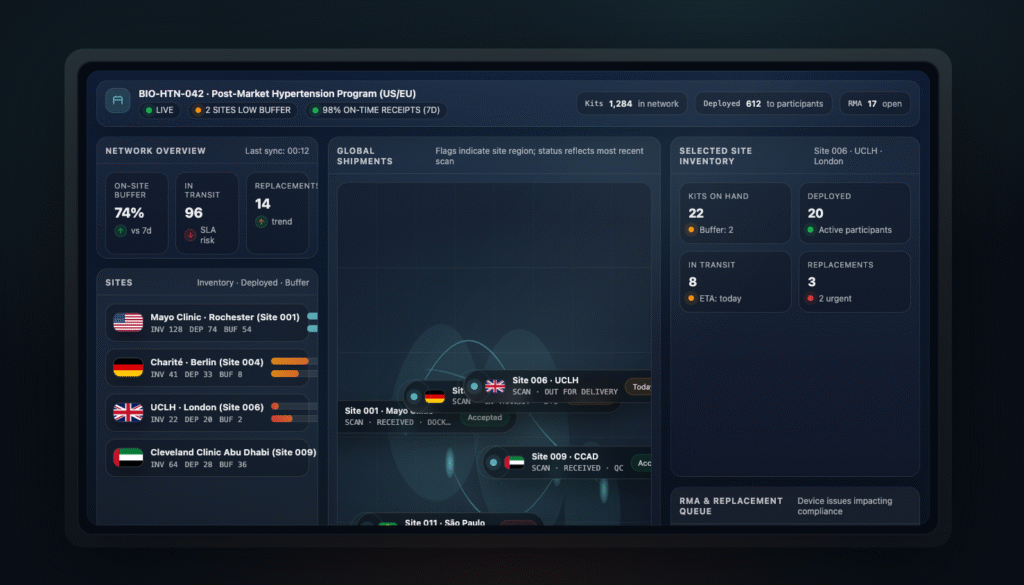
Every device — from tablet to wearable to charger — is tracked in Delve’s supply chain system. Study teams always know what’s deployed, what’s in transit, and what needs replacement.
No more guesswork. No more spreadsheets. True supply chain intelligence.
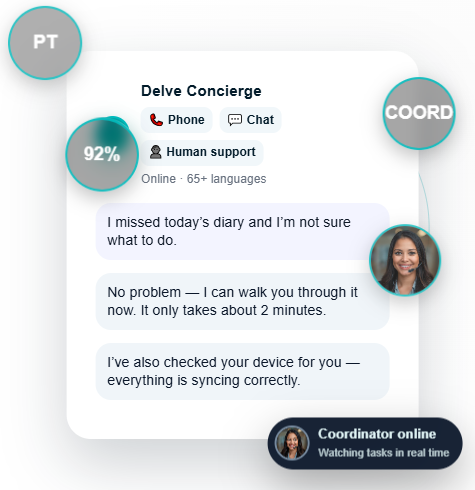
Logistics is only half the equation. Devices still fail, batteries die, and patients need help — which is why Delve pairs provisioning with live multilingual concierge support.
This combination produces unmatched device compliance.

Let Delve handle the complexity — sourcing, shipping, setup, support, and returns — so your teams can focus on science.