eCOA completion
92–98%
Typical diary completion
Retention uplift
+40%
With layered concierge support
Global readiness
120+
Languages supported
For pharma teams running global trials
Unified eCOA, validated wearable endpoints, and protocol-trained concierge support help pharma teams run decentralized and hybrid trials with confidence—while reducing site burden and preventing deviations early.
Cleaner evidence · Fewer missed tasks · Faster issue resolution.
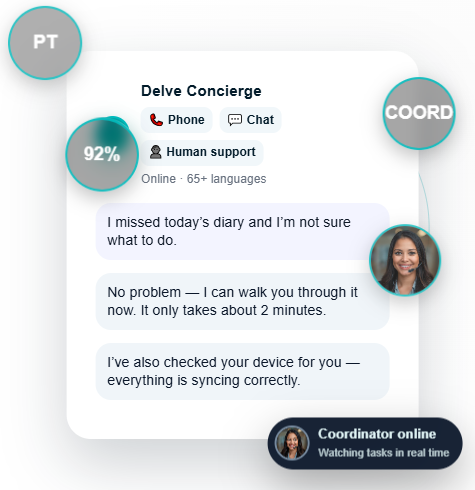
Today’s trials generate millions of signals — but only actionable, validated endpoints move programs forward. Delve Health brings eCOA, wearables, and multilingual concierge together into a single, regulatory-aligned ecosystem.

Deploy globally, validate confidently, and keep evidence flowing—Delve combines
eCOA, digital measures, consent, and analytics with a human layer that prevents drift
and missed data.
Deploy multilingual eCOA + wearables across any region with consistent operations.
Fit-for-purpose validation and documentation designed for FDA/EMA review expectations.
Real humans intervene when patients fall behind—recovering syncs and preventing deviations.
Live dashboards for safety signals, sync failures, and endpoint quality—without waiting for exports.
Instead of juggling siloed vendors, Delve harmonizes subjective and objective data streams into a single real-time environment.

When a device misses a sync, a diary goes incomplete, or a patient feels lost — automation flags the issue, and our concierge team resolves it in minutes.

95% retention at 12 months with concierge + wearable oversight.

Objective signals correlated with gold-standard clinical measures.

63% improvement in adherence through patient outreach programs.
Device Compliance
Retention at 12 Months
Fewer Missed Syncs
Faster Enrollment Support
Real-time oversight. Validated digital endpoints. Human-centered retention.