eCOA completion
92–98%
Typical diary completion
Real-time QC
< 24h
Query + deviation signals
Study activation
Days
Configured, tested, deployed
eCOA built for real-world research
Delve Health combines modern eCOA with real-time QC, wearable data, and human support to keep diaries complete, reduce deviations, and eliminate site troubleshooting burden.
92–98% diary completion · 2–3× retention uplift.
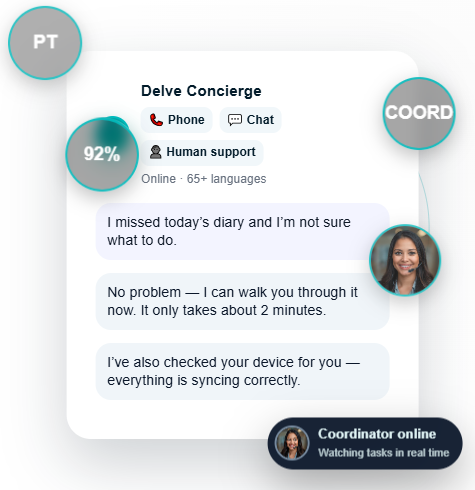
Clinical outcomes fail when patients miss diaries, devices break, or sites can’t support troubleshooting. Delve Health solves this by blending modern eCOA, wearable-driven insights, and human engagement into one unified ecosystem.
Data becomes more reliable. Patients stay supported. Sites stay unburdened. Sponsors get cleaner evidence with fewer deviations and faster readouts.

High-performing eCOA isn’t just digital forms. It’s the intersection of patient experience,
protocol rigor, and continuous support designed to work in the real world.
Mobile-first assessments with adaptive UX, multimedia prompts, and multilingual support.
Validated, fit-for-purpose measures from wearables and sensors, ready for regulators.
Human teams who own the work of keeping participants active, informed, and on schedule.
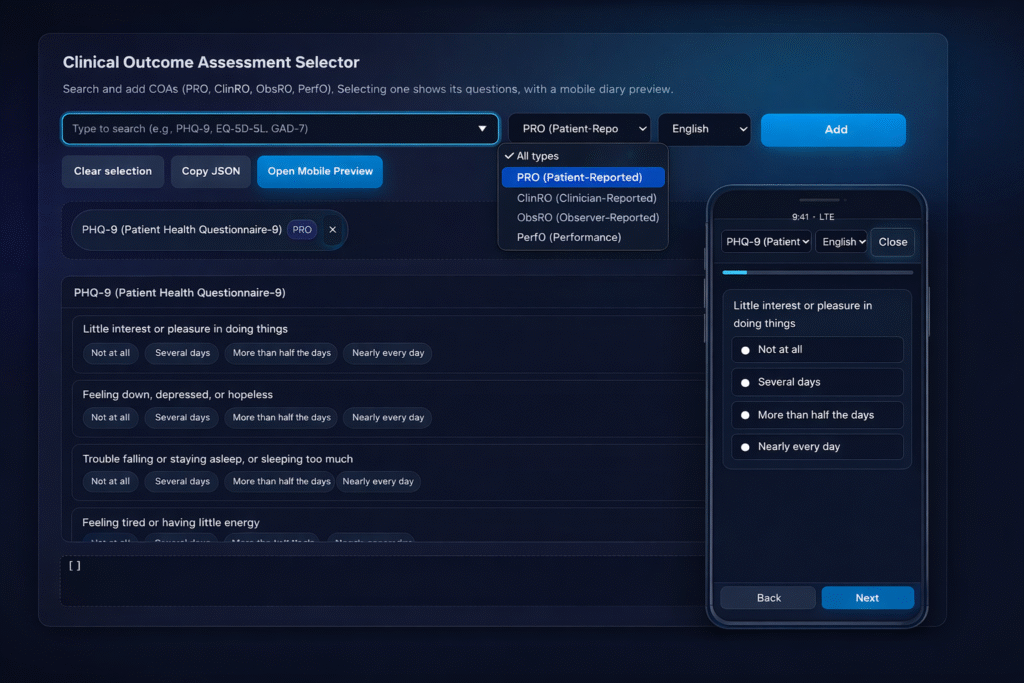
Build protocol-aligned instruments in minutes — no custom development needed. StudyPal’s authoring tools let research teams design, test, validate, and deploy assessments faster than traditional eCOA vendors.
All forms stay aligned with regulatory expectations — including FDA, EMA, and ICH E6(R3) guidance.
Wearables provide objective data; ePRO captures subjective experience. Delve Health merges the two — enabling richer endpoints, early signal detection, and context around patient behavior.
This is the future of evidence generation: signals + symptoms → complete insight.

Software alone cannot fix missed diaries, low tech literacy, battery issues, or sync failures. Delve’s Concierge-as-a-Service™ becomes the operational engine behind your study.
It’s not just eCOA — it’s eCOA with a human safety net.


92% ePRO completion using concierge follow-up on missed tasks.
45% improvement in daily symptom diary adherence.
30% faster interim analysis using unified ePRO + wearable data.
ePRO Completion
Improved Daily Adherence
Faster Data Availability
Languages Supported
Unified assessments. Wearable-connected endpoints. Human engagement behind every data point.